Research in the Gu Lab
The basal ganglia circuits are essential for everyday behavioral and physiological functions, and pathological alterations within these circuits lead to profound impairments in neurological disorders. The Gu Laboratory is dedicated to unraveling how basal ganglia dysfunctions drive behavioral and physiological abnormalities, with a particular emphasis on movement disorders. To address this goal, the laboratory investigates the circuit mechanisms underlying diverse forms of behavioral control, while also examining the role of the basal ganglia in regulating physiological processes such as respiration.
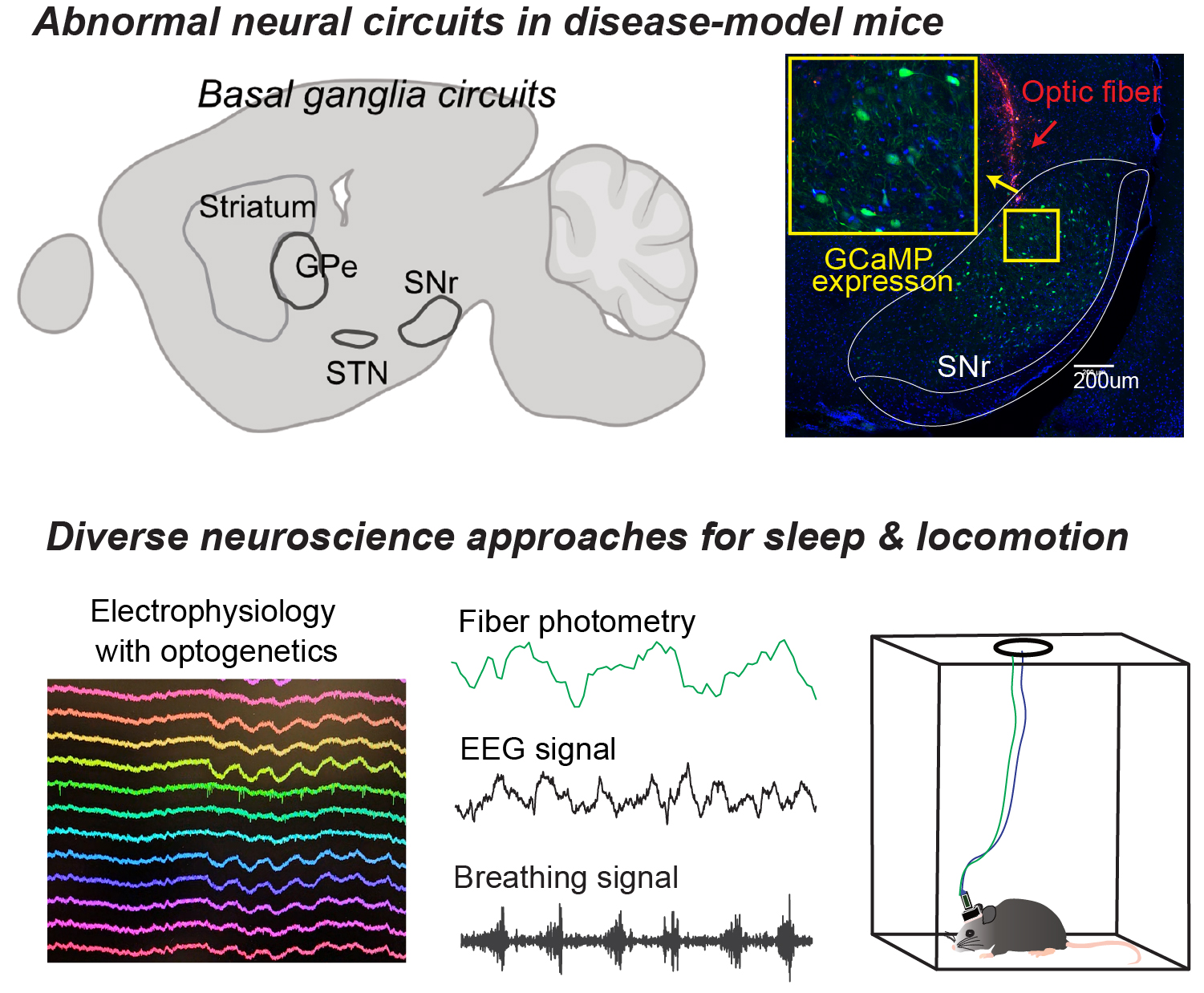
Because respiratory dynamics are closely linked to behavioral states including sleep and locomotion, investigating basal ganglia contributions to both behavioral and physiological regulation provides a novel framework for understanding brain-body interactions. By integrating these dimensions, the Gu Lab aims to reveal how neural circuit dysfunction disrupts coordinated behavioral and physiological outputs in health and disease. This approach elucidates general principles by which basal ganglia circuits integrate neural activity with internal physiological states to shape behavior. By combining circuit-level analysis with physiological monitoring across behavioral contexts, the lab seeks to establish a unified framework for understanding how brain circuits coordinate action, arousal, and bodily function. These efforts not only advance fundamental knowledge of basal ganglia organization but also provide a foundation for understanding how disruptions in brain circuit–physiology coupling contribute to diverse neurological and neuropsychiatric conditions.
To achieve these goals, the Gu Lab applies a broad array of modern neuroscience methodologies in behaving mice to investigate basal ganglia circuit dynamics. Using in vivo electrophysiology and fiber photometry, the lab measures neural activity across behavioral states to define the functional roles of specific basal ganglia cell types. In particular, Dr. Gu has advanced the use of neural population dynamics analyses to capture complex, high-dimensional neural activity in behaviorally relevant spaces. In addition, the lab uses cell-type-specific optogenetic manipulations to establish causal relationships between neural activity and behavior.
Moreover, the Gu Lab simultaneously records respiration and cardiac signals alongside neural activity, enabling precise examination of neural-physiological interactions during behaviors – an area that has been largely underexplored in the field. By integrating these approaches, the lab investigates how basal ganglia circuits interact with respiratory control systems across behavioral state changes such as sleep and locomotion.
In parallel, the Gu Lab examines these processes in disease-relevant mouse models to identify pathological alterations in circuit-physiology interactions. A major focus is on Parkinson’s disease models, with the goal of uncovering the neural and physiological mechanisms underlying abnormal locomotion and sleep disturbances commonly observed in patients. By systematically addressing these motor and non-motor symptoms, the lab seeks to identify precise circuit mechanisms and develop strategies to more effectively alleviate these impairments.
Through collaborations with clinical centers within the Hackensack Meridian Health network, the Gu lab aims to bridge basic and clinical research to advance understanding of basal ganglia dysfunction and to lay the foundation for the development of targeted, circuit-based therapeutic approaches for neurological disorders. By fostering close integration between basic and clinical neuroscience, these efforts support the development of mechanistically informed interventions and future translational research initiatives.
Selected Publications
- Gu BM, Schmidt R, Berke JD (2020) Globus pallidus dynamics reveal covert strategies for behavioral inhibition. eLife 9:e57215. PMID: 32519952
- Gu BM†, Berke JD (2022) Altered basal ganglia output during self-restraint. eLife. 11:e82143. PMID: 36321810
- Gu BM*, Kim JG*, Hossain A, Cron GO, Lee JH (2025) Substantia nigra modulates breathing rate via locus coeruleus. iScience. 28(5):112423. PMID: 40343266
- Dehdar K, Neuberg E, Gu BM† (2025) Dynamic respiration-neural coupling in substantia nigra across sleep and anesthesia. J Neurosci. 2025 Nov 17:e1154252025. PMID: 41249057
†Corresponding author *Co-first authors
