Research in the Nagajyothi Lab
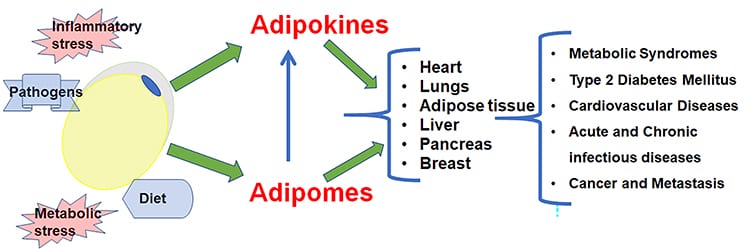
Our laboratory has focused on investigating: (i) the role that adipocytes play in the immune and metabolic systems and (ii) the effect of dysfunctional adipocytes in the pathogenesis, progression and severity of various infectious and non-infectious diseases. Adipocytes are the major regulator of whole-body energy and immune homeostasis. We have demonstrated that adipocytes themselves are a source of inflammatory mediators (IL-6, IL-10, IL1β, IFN-γ, TNF-α, CCL2, CCL5, CXCL10, leptin, and adiponectin). Many factors including infections, diseases, diet, and exercise influence adipocyte physiology, while the pathology of adipocytes influences the outcome of diseases. Our laboratory is currently examining the crosstalk between the adipomes (extracellular vesicles) released by pathogenic adipocytes and the outcome of diseases in the research areas described below.
Impact of the loss of adipocytes on the progression and severity of acute and chronic infectious diseases
We use mouse models of acute and chronic infectious diseases, Chagas cardiomyopathy and pulmonary tuberculosis (TB), caused by the parasite Trypanosoma cruzi and bacterium Mycobacterium tuberculosis, respectively, to examine the impact of gain or loss of body fat mass and fat cells on disease outcomes. Our earlier studies demonstrated a correlation between increased body fat mass and increased survival during acute infection, whereas loss of fat mass correlated with faster Disease progression and increased severity during the chronic stage of infection. We recently discovered that adipocytes release adipomes, which are specific to the pathogenic condition of the organism, and which regulate other cells, including immune cells. Currently, our studies are focused on investigating disease associated immune-metabolic pathways, which are regulated by pathogenic adipomes via their specific cargoes, to identify specific drug targets to prevent the progression of Chagas cardiomyopathy and activation/reactivation of TB disease.
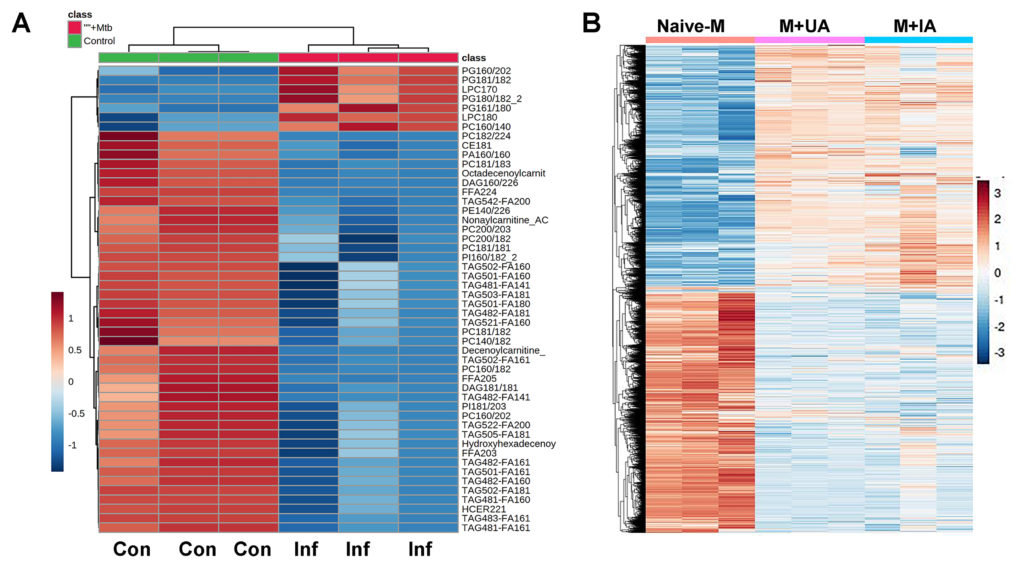
Adipomes carry cargoes specific to the pathological conditions in adipose tissue and can regulate cells in proximity and distant sites.
A: Representative heat-maps showing a diverse lipid profile in adipomes isolated between, uninfected control and Mycobacterium tuberculosis aerosol infected mice (30 days post-infection).
B: Representative heatmap showing differentially expressed genes in Raw264 cells incubated with adipomes isolated from uninfected (M+UA) and acute Trypanosoma cruzi infected mice (M+IA) compared to Naïve macrophages as demonstrated by RNA seq analysis.
Role of pathogenic adipocytes in the tumorigenesis and metastasis of breast cancer
Obesity is a known risk factor for the development of breast cancer and in promoting metastasis in post-menopausal women, whereas obesity reduces the risk of breast cancer in pre-menopausal women. The female breast stroma is mostly made up of adipose tissue containing mainly mature adipocytes, preadipocytes, and adipose tissue derived stem cells (ASC). Breast tumor progression also depends on the crosstalk between tumor epithelial cells and the surrounding adipocytes. Our lab is studying the role of pathogenic adipocyte-derived adipomes in regulating tumor associated macrophages and in promoting tumor cell growth and metastasis. We are collaborating with Drs. Joyce Slingerland, Sonia de-Assis, and Olivier Loudig to investigate the regulatory roles of adipomes in the tumor microenvironment and the systemic macroenvironment, especially focusing on adipome effects on tumor progression, distant metastasis, and therapeutic resistance.
Influence of diet on the overactivated immune system during acute infections
The physiology of each organ depends on its metabolic status, and the metabolic requirements of the heart, lung, brain, adipose tissue, muscle, kidney, and liver are strikingly different. Most localized acute infections affect the metabolic status of both the infected organ and the whole organism. The activity of immune cells is regulated by the metabolic environment. Our recent studies demonstrated that a high-fat diet reduces the levels of pro-inflammatory signaling in the infected area, whereas a carbohydrate-rich diet increases the levels of pro-inflammatory mediators in murine models of acute Chagas and TB diseases. We are currently investigating the effect of diets on the pathophysiology of adipocytes and on regulating the activity of localized and systemic immune cells.
Effect of metabolic syndromes on infectious diseases and vice versa
The term “metabolic syndrome” describes a group of risk factors for the development of diabetes and cardiovascular disease. Currently, the incidence of metabolic syndromes is increasing worldwide while the incidence of old infectious diseases is decreasing due to the available drugs and vaccines. However, recent clinical and research data have shown that subjects with metabolic syndromes are particularly vulnerable to acute and chronic infectious diseases. Our lab investigates the role of adipocytes in the progression of metabolic syndromes and their effect on the outcomes of infectious diseases such as Chagas and TB diseases. Currently we are aiming to investigate the role of pathogenic adipocytes in the severity of COVID-19 patients with metabolic syndromes, T2DM and cardiovascular diseases. In addition, our laboratory is also investigating whether, conversely, these infectious diseases contribute to the pathogenesis of metabolic syndromes. Our results show a strong link between asymptomatic infections, anti-TB drug treatments, and the development of insulin resistance. We are investigating the effect of either acute or chronic presence of infectious microbes in AT and the effects of antibiotic treatment on adipocyte physiology and on the pathogenesis of metabolic syndrome and the development of metabolic disorders.
The overarching goal of our research is to demonstrate that adipocyte-derived adipokines and adipomes could serve as therapeutic targets for various infectious and non-infectious diseases.
