iMML’s Mission
The Institute for Multiple Myeloma and Lymphoma (iMML) is evolving to represent a true “clinical laboratory” for multiple myeloma and lymphoma, including real world research-grade data over time, and access to clinical material over the continuum of care. In collaboration with the John Theurer Cancer Center, home to one of the best and most active clinical multiple myeloma programs in the world, the Institute will test every legitimate therapeutic option available and become one of the most consistent conduits of drug development in myeloma. We seek to transform the treatment of patients with multiple myeloma by integrating our enormous clinical expertise and research contributions with ongoing basic and translational research.
Collaborative Framework
The landscape of partnerships in biomedical research and development is rapidly evolving. The iMML, supported by a major philanthropic investment in 2017 by a long-time pharmaceutical partner, is a combination of a large institute, academic drug discovery center, and risk-sharing model, funded through philanthropic investment and future commercialization initiatives. Our decades-long industry collaboration, strengthened by the transformational gift, will benefit patients and their families, scientists, and physicians, while also spurring economic development in northern New Jersey – all stemming from the joint commitment to cure multiple myeloma. The iMML will hasten the development of breakthrough ideas to their full expression in clinical trials and approved interventions, ultimately offering life-saving care for patients.
Our Approach
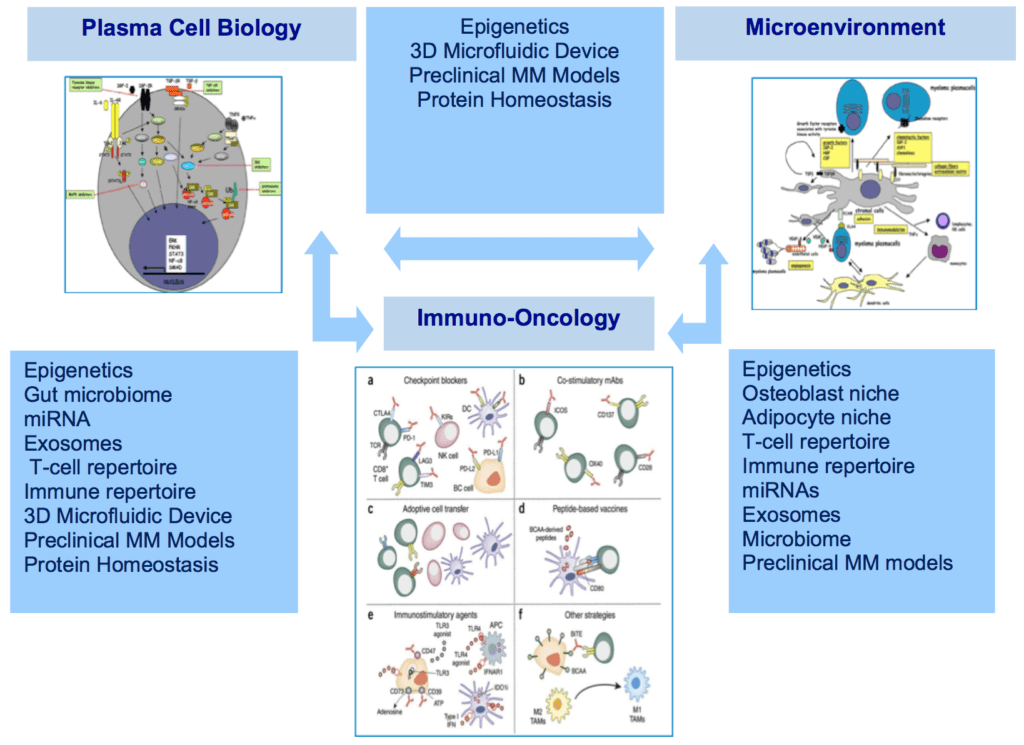
While we have more genomic information about multiple myeloma than virtually any other neoplastic diseases, this knowledge has not yet been translated into more effective therapies. Numerous studies have highlighted the biological complexity of multiple myeloma that arises as a result of several disrupted cancer pathways. In addition, the disease shows great heterogeneity, reflected by its wide panel of genetic alterations and the individual differences in overall response and survival of patients receiving the same treatment. In fact, we have not identified master “driver” mutations or thematic pathways shared across all cases, and there is no actionable genetic signature for multiple myeloma. Epigenetics represents the system that controls gene expression (i.e. turns them on or off). Recent work suggest that epigenetics changes observed in myeloma cells are critical for response or resistance to therapy, and drugs that alter epigenetic marks on the DNA are being pursued.
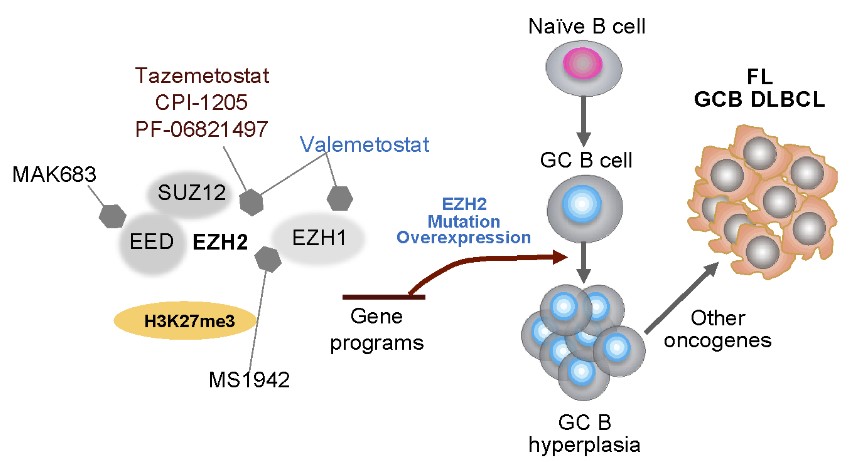
At the forefront of medical research, the iMML is dedicated to unraveling the intricate complexities of lymphoma. One of our pioneering research programs investigates the role of EZH2, a chromatin-modifying enzyme, which is critical for cell proliferation, differentiation, expansion, and function. EZH2 is involved in B cell lymphomagenesis, including follicular lymphomas (FLs) and germinal center B (GCB) cell type diffuse large B-cell lymphomas (DLBCLs). Gain-of-function EZH2 mutations in GCB cells suppress expression of genes involved in terminal differentiation and proliferation checkpoints, leading to persistent centroblast proliferation and GC hyperplasia. The high incidence of EZH2 mutations in lymphomas and their stability during disease progression makes EZH2 an effective target to develop drugs for a targeted therapy.
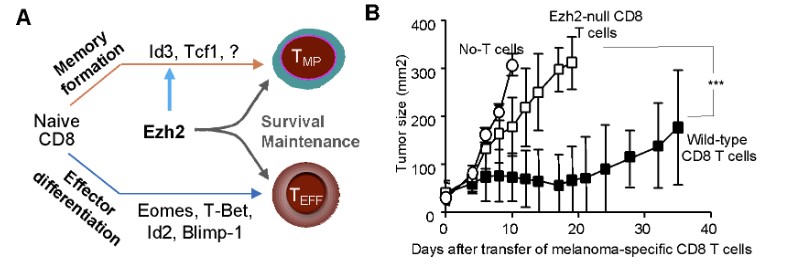
In a recent study, our research team discovered that EZH2 plays a central role in regulating T cell immunity against tumor. EZH2 restrains development of terminal differentiation of effector CD8 T cells, promotes formation of precursor and mature memory CD8 T cells, regulates appropriate specification and identity of helper CD4 T cells, and maintains survival of differentiated antigen-specific T cells. Most importantly, EZH2 is shown to be important for reinvigoration of exhausted chimeric-antigen-receptor (CAR)-T cells. Led by our esteemed experts, the members of iMML are passionately committed to advancing our understanding of EZH2 regulation of T cell immunity for innovative therapies by identifying and exploring novel targets such as EZH2. Through meticulous study and collaborative efforts, we are driven by the hope of transforming the landscape of lymphoma treatment and offering renewed possibilities for patients.
Scientific Approach
For decades, the study of the complex biology of multiple myeloma and lymphoma has consumed scientists and physicians in the field. The cure for multiple myeloma and lymphoma – and ultimately many other cancers – will be discovered through a multipronged and sequential approach to achieve the following:
- Direct eradication of cancer cells
- Manipulation of the microenvironment (removal of hiding niches) to preclude their return
- Strengthening the immune response to prevent recurrence and maintain a disease-free state (immuno-oncology)
The link between the cellular microenvironment and effective and continuous immune surveillance appears now to hold the key to a cure for multiple myeloma and other related blood disorders.
iMML Team
David Siegel, M.D.
Director, iMML
Chief and Founder of the Multiple Myeloma Division
John Theurer Cancer Center, Hackensack Meridian Health
davids.siegel@hmhn.org
Rena Feinman, Ph.D.
Member, iMML
rena.feinman@hmh-cdi.org
Benjamin Tycko, M.D., Ph.D.
Member, iMML
benjamin.tycko@hmh-cdi.org
Johannes Zakrzewski, M.D..
Member, iMML
Johannes.Zakrzewski@hmh-cdi.org
Yi Zhang, M.D., Ph.D.
Member, iMML
yi.zhang@hmh-cdi.org
Hai-Hui (Howard) Xue, M.D., Ph.D.
Member, iMML
haihui.xue@hmh-cdi.org
Physician Scientists at Hackensack Meridian Health
Andre Goy, M.D.
Chairman & Chief Physician Officer
Lymphoma Division Chief
John Theurer Cancer Center
Hackensack University Medical Center
goy.andre@hmhn.org
Michael D. Stifelman, M.D.
Chair of Urology and Director of Robotic Surgery, Hackensack University Medical Center
Professor and Founding Chair of Urology, Hackensack Meridian School of Medicine
George John Kaptain, M.D.
Surgical Director of Neuro-Oncology
Skull-Base Surgery and Radiosurgery
John Theurer Cancer Center,
Hackensack University Medical Center
Lombardi Comprehensive Cancer Center - Based Faculty Members
Louis Weiner, M.D.
Director, Georgetown Lombardi Comprehensive Cancer Center
Professor and Chair,Department of Oncology, Georgetown University Medical Center.
weinerl@georgetown.edu
